ASPP2, a New Regulator of the Mevalonate Pathway in Cancer
Beibei Liang1,2, Yangyang Xie1, Lisha Zhang1,3, Jian Zhao1,3*
1Shanghai Key Laboratory of Molecular Imaging, Shanghai University of Medicine & Health Sciences, Shanghai, 201318, China
2Shanghai Key Laboratory of Molecular Imaging, Jiading District Central Hospital Affiliated Shanghai University of Medicine and Health Sciences, Shanghai 201318, China
3Shanghai University of Traditional Medicine, Shanghai 201203, China
Tumor is the abnormal growth tissue, which needs adequate energy and abundant material to meet requirement for growth. Cholesterol is an essential lipid to maintain intracellular homeostasis. It is an important part of the cell membrane and a primer precursor of steroids and vitamin D. The mevalonate (MVA) pathway is a metabolic pathway for the synthesis of isoprene pyrophosphate and daily pyrophosphate from acetyl-CoA, which regulates the biosynthesis of cholesterol and non-sterol isoprene. Isoprene is used to synthesize important biomolecules in vivo, such as dodecyl, heme A, ubiquinone, coenzyme Q, and hydrophobic long chains, the last one can anchor proteins such as Ras and Rho to the cell membrane for signal transduction. Therefore, mevalonate pathway is related to a variety of cell processes, involved in cell proliferation, differentiation, tumorigenesis, metastasis, and immune escape1.
In recent years, a large number of studies have shown that the metabolites of MVA pathway are beneficial to the survival of tumor cells, and a variety of key enzymes of MVA pathway are essential for the survival of tumor cells2-4. Tumor cells need sufficient cholesterol and intermediates of the cholesterol biosynthesis to maintain their high levels of proliferation.
Transcriptional Regulation of the Mevalonate Pathway
Transcriptional activation of key enzymes in MVA pathway, like 3-hydroxy-3-methylglutaryl-CoA synthase 1 (HMGCS1), 3-hydroxy-3- methylglutaryl-CoA reductase (HMGCR) are controlled by sterol regulated element binding protein-2 (SREBP-2). SREBP2, a member of SREBPs family, which is synthesized as an inactive precursor locating on endoplasmic reticulum (ER) membrane5. The activation of SREBP2 is normally regulated by cholesterol level in ER, upon cholesterol level decrease SREBP-2 is translocated from ER to the Golgi apparatus, leading to subsequent proteolytic cleavage to its active mature form6. Mature SREBP-2 translocates to nucleus and binds with sterol regulatory elements (SREs) in the promoters of its target genes to regulate MVA pathway and cholesterol synthesis7.
Recent studies have shown that the transcriptional activity of SREBP-2 can be regulated by several tumor suppressor genes or oncogenes8. Tumor suppressor gene p53 plays an important regulatory role in the tumors progression. Studies have confirmed that the MVA pathway is a new way for p53 to inhibit tumor growth. P53 blocks the activation of SREBP-2 by inducing the expression of ABCA1, a member of ATP-binding cassette (ABC) transporters which regulate cholesterol transport across membrane. Derepression of the MVA pathway is essential for tumorigenesis caused by p53 loss in Myc induced mouse liver cancer9. Mutant TP53 (TP53R273H and TP53R280K) can bind to SREBP-2 and increase the transcription of MVA pathway genes, which contributes to mutant p53 induced disruption of normal breast acinar morph10. Mutant p53 can also up-regulate the activity of MVA pathway-related oncogene (such as RAS, Rho, YAP/TAZ), thus promoting tumor progression11.Rb prevents SREBP-2 from being associated with the target gene promoter and acts as a negative regulator of MVA metabolism12. Data from the Encyclopedia of DNA Elements (ENCODE) project has shown that MYC binds to promoters of MVA pathway genes in close proximity to SREBP-2, which may contribute to Myc-induced hepatocarcinogenesis12. YAP target gene ZMYND8 interacts with SERBP2 to up-regulate the MVA pathway13. HSP90 and the MVA pathway promote STAT3 phosphorylation mediated by mutant p53 in glioblastoma and pancreatic cancer cells14. PI3K/AKT signaling activates the MVA pathway through increasing the expression of SREBPs15. Schematic representation summarizes the most important regulation of SREBP2 in the MVA Pathway (Fig. 1).
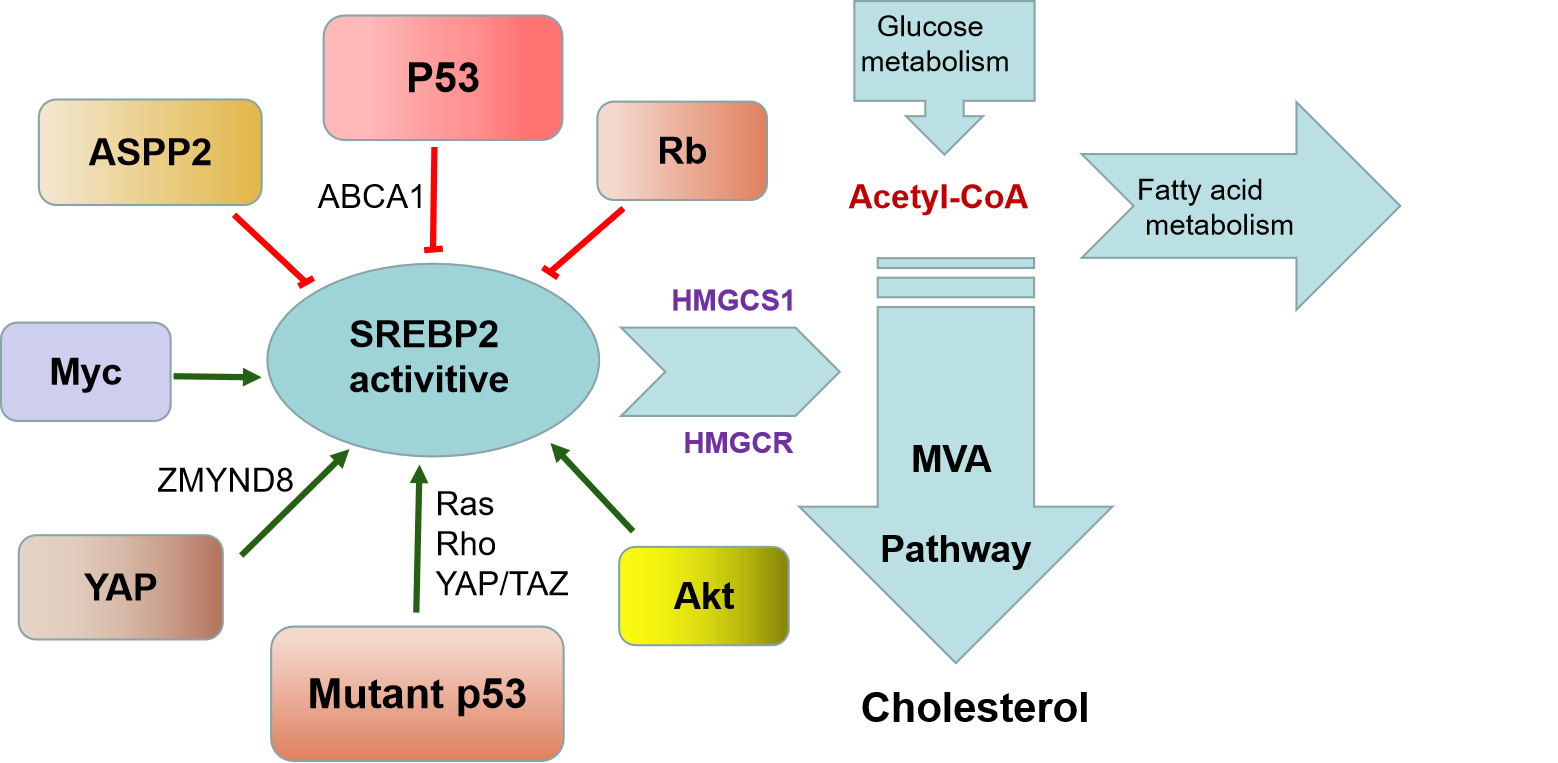
Figure 1: Regulation of SREBP2 in the MVA Pathway
ASPP2, a New Regulator of SREBP2 and the MVA Pathway
ASPP2 (Apoptosis-stimulating protein of p53) is a member of the ASPP family. ASPP2 is identified as an activator of the p53 family, which can promote apoptosis16. In human cancer, ASPP2 expression is down-regulated epigenetically by DNA methylation or histone deacetylation17,18. As we know, a large number of studies have confirmed that ASPP2 can regulate a series of biological process by cooperating with specific targets, such as p73, Bcl-2, NF-kB, Yes-Association Protein-1, RAS, Par3, β-catenin, and Beclin-1. Most of the previous studies suggest that ASPP2 is mainly expressed in tumor cytoplasm or cell membrane. ASPP2 expressed on the cell membrane can bind to PAR3 through its N-terminal to maintain the integrity of cell polarization19,20, and can bind to β-catenin-E-cadherin complex to regulate epithelial plasticity21. In the cytoplasm, ASPP2 binds to RAS to regulate RAS signaling pathway22,23 and interacts with ATG5 and Beclin-1 to regulate autophagy24,25. However, nucleic ASPP2 is important for it to execute its proapoptotic effect. In the nucleus, ASPP2 binds to p53 and p73 through its C-terminal26 and function as a transcription cofactor to regulate p53 target apoptotic genes. Whether ASPP2 also plays a role in the MVA pathway? That attracted us more interests in recent years. In our previous study, we found ASPP2 interacted with SREBP-2 in the nucleus and negatively regulated SREBP-2 transcriptional activity and its target MVA pathway genes27. HCC tissues with nucleic ASPP2 staining were more prone to have low HMGCR expression than HCC tissue with cytoplasmic ASPP2 staining, further supporting a negative effect of nuclear ASPP2 on the MVA pathway. Given that p53 and RAS are essential regulators in MVApathway, whether the interactions between ASPP2 and p53, ASPP2 and RAS, crosstalk in MVA pathway still need further investigation. The key role of ASPP2 in regulating MVA metabolism and its importance in tumor growth may provide a new strategy for the treatment of cancer (Fig. 2).
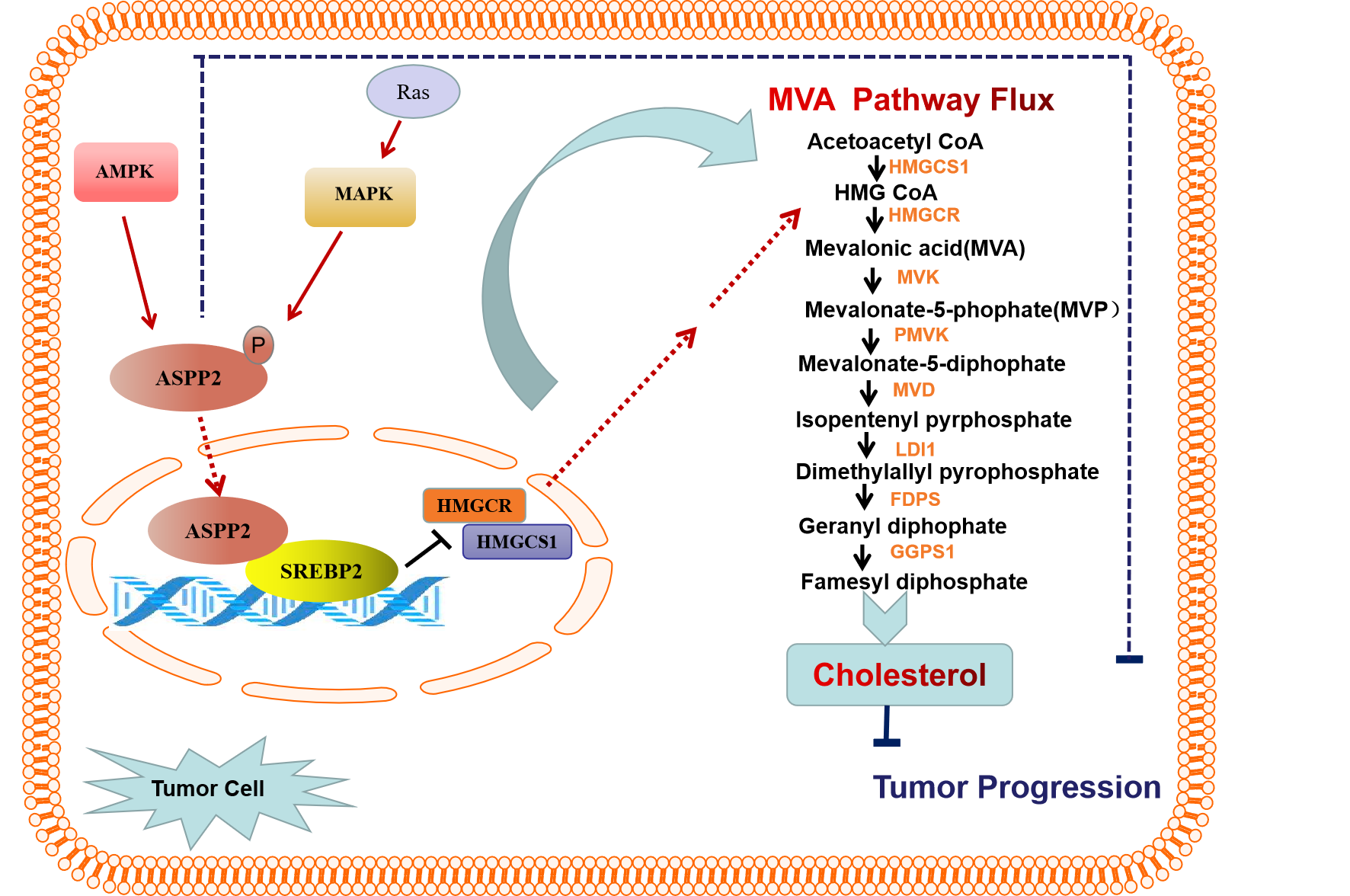
Figure 2: ASPP2 inhibits tumor growth by repressing the MVA pathway
The mechanism by which ASPP2 translocates to nucleus is still unclear. There are Ankyrin repetitive sequences at the C terminal of ASPP2, which may help ASPP2 enter the nucleus through the establishment of RanGDP/Ankyrin binding nuclear input pathway28. Phosphorylation of ASPP2 may be necessary for it to enter into nuclear. Phosphorylation of ASPP2 protein at Ser827 by mitogen-activated protein kinase pathway can cause ASPP2 translocation from the cell membrane to the nucleus, which is critical for its proapoptotic effect22,29. In a system-wide phosphoproteome study, AMP-activated protein kinase phosphorylates ASPP2 at Ser479 to promote apoptosis in an irradiation-dependent way30. In addition, nuclear translocation of ASPP2 may related to inflammation stimulation in immune cells. The transfer of ASPP2 from the cytoplasm to the nucleus in mouse macrophages is found in the mouse model of maternal inflammation induced by LPS31.
Conclusions and Future Perspectives
Metabolic reprogramming is a hallmark of tumor cells. The MVA pathway recently has been regarded as an attractive target for tumor therapy. As a haplo-insufficient tumor suppressor, ASPP2 can be expressed in tumor cytoplasm, cell membrane and nuclear. Nucleic ASPP2 has been found to inhibit cholesterol biosynthesis by regulating the MVA pathway. But what stimulates ASPP2 to transfer from cytoplasm to nuclear? Whether nuclear ASPP2 has more functions in tumor progression deserves our further attention.
References
- Mullen PJ, Yu R, Longo J, et al. The interplay between cell signalling and the mevalonate pathway in cancer[J]. NAT REV CANCER, 2016, 16(11): 718.
- Hart T, Chandrashekhar M, Aregger M, et al. High-resolution CRISPR screens reveal fitness genes and genotype-specific cancer liabilities[J]. CELL, 2015, 163(6): 1515.
- 3. Wang T, Birsoy K, Hughes NW, et al. Identification and characterization of essential genes in the human genome[J]. SCIENCE, 2015, 350(6264): 1096.
- Blomen VA, Májek P, Jae LT, et al.Gene essentiality and synthetic lethality in haploid human cells[J]. SCIENCE, 2015, 350(6264): 1092.
- Nohturfft A, Zhang SC. Coordination of lipid metabolism in membrane biogenesis[J]. Annual Review of Cell and Developmental, 2009, 25: 539.
- Yang T, Espenshade PJ, Wright ME, et al. Crucial step in cholesterol homeostasis: sterols promote binding of SCAP to INSIG-1, a membrane protein that facilitates retention of SREBPs in ER[J]. CELL, 2002, 110(4): 489.
- Amemiya-Kudo M, Shimano H, Hasty AH, et al. Transcriptional activities of nuclear SREBP-1a,-1c, and-2 to different target promoters of lipogenic and cholesterogenic genes[J]. J LIPID RES, 2002, 43(8): 1220.
- Mullen PJ, Yu R, Longo J, et al. The interplay between cell signalling and the mevalonate pathway in cancer[J]. NAT REV CANCER, 2016, 16(11): 718.
- Moon S, Huang C, Houlihan SL, et al. p53 represses the mevalonate pathway to mediate tumor suppression[J]. CELL, 2019, 176(3): 564.
- Freed-Pastor WA, Mizuno H, Zhao X, et al. Mutant p53 disrupts mammary tissue architecture via the mevalonate pathway[J]. CELL, 2012, 148(1-2): 244.
- Parrales A, Thoenen E, Iwakuma T. The interplay between mutant p53 and the mevalonate pathway[J]. Cell Death & Differentiation, 2018, 25(3): 460.
- Shamma A, Takegami Y, Miki T, et al.Rb Regulates DNA damage response and cellular senescence through E2F-dependent suppression of N-ras isoprenylation[J]. CANCER CELL, 2009, 15(4): 255.
- Pan Q, Zhong S, Wang H, et al. The ZMYND8-regulated mevalonate pathway endows YAP-high intestinal cancer with metabolic vulnerability[J]. MOL CELL, 2021.
- 14. Romeo MA, Gilardini Montani MS, Benedetti R, et al. STAT3 and mutp53 engage a positive feedback loop involving HSP90 and the mevalonate pathway[J]. FRONT ONCOL, 2020, 10: 1102.
- Yi J, Zhu J, Wu J, et al. Oncogenic activation of PI3K-AKT-mTOR signaling suppresses ferroptosis via SREBP-mediated lipogenesis[J]. Proceedings of the National Academy of Sciences, 2020, 117(49): 31189.
- Bergamaschi D, Samuels Y, Jin B, et al. ASPP1 and ASPP2: common activators of p53 family members[J]. MOL CELL BIOL, 2004, 24(3): 1341.
- Zhao J, Wu G, Bu F, et al. Epigenetic silence of ankyrinârepeat–containing, SH3âdomain–containing, and prolineârichâregion–containing protein 1 (ASPP1) and ASPP2 genes promotes tumor growth in hepatitis B virus–positive hepatocellular carcinoma[J]. HEPATOLOGY, 2010, 51(1): 142.
- Li H, Wang X, Zhang C, et al. HDAC1-induced epigenetic silencing of ASPP2 promotes cell motility, tumour growth and drug resistance in renal cell carcinoma[J]. CANCER LETT, 2018, 432: 121.
- Sottocornola R, Royer C, Vives V, et al. ASPP2 binds Par-3 and controls the polarity and proliferation of neural progenitors during CNS development[J]. DEV CELL, 2010, 19(1): 126.
- Cong W, Hirose T, Harita Y, et al. ASPP2 regulates epithelial cell polarity through the PAR complex[J]. CURR BIOL, 2010, 20(15): 1408.
- Wang Y, Bu F, Royer C, et al. ASPP2 controls epithelial plasticity and inhibits metastasis through β-catenin-dependent regulation of ZEB1[J]. NAT CELL BIOL, 2014, 16(11): 1092.
- Wang Y, Godin-Heymann N, Wang XD, et al. ASPP1 and ASPP2 bind active RAS, potentiate RAS signalling and enhance p53 activity in cancer cells[J]. Cell Death & Differentiation, 2013, 20(4): 525.
- Wang Z, Liu Y, Takahashi M, et al. N terminus of ASPP2 binds to Ras and enhances Ras/Raf/MEK/ERK activation to promote oncogene-induced senescence[J]. Proceedings of the National Academy of Sciences, 2013, 110(1): 312.
- Wang Y, Wang XD, Lapi E, et al. Autophagic activity dictates the cellular response to oncogenic RAS[J]. Proceedings of the National Academy of Sciences, 2012, 109(33): 13325.
- Chen R, Wang H, Liang B, et al. Downregulation of ASPP2 improves hepatocellular carcinoma cells survival via promoting BECN1-dependent autophagy initiation[J]. CELL DEATH DIS, 2016, 7(12): e2512.
- Trigiante G, Lu X. ASPPs and cancer[J]. NAT REV CANCER, 2006, 6(3): 217.
- Liang B, Chen R, Song S, et al. ASPP2 inhibits tumor growth by repressing the mevalonate pathway in hepatocellular carcinoma[J]. CELL DEATH DIS, 2019, 10(11): 1.
- Lu M, Zak J, Chen S, et al. A code for RanGDP binding in ankyrin repeats defines a nuclear import pathway[J]. CELL, 2014, 157(5): 1130.
- Godin-Heymann N, Wang Y, Slee E, et al. Phosphorylation of ASPP2 by RAS/MAPK pathway is critical for its full pro-apoptotic function[J]. PLOS ONE, 2013, 8(12): e82022.
- Jiang Y, Cong X, Jiang S, et al. Phosphoproteomics reveals AMPK substrate network in response to DNA damage and histone acetylation[J]. Genomics, proteomics & bioinformatics, 2021.
- Turnquist C, Wang Y, Severson DT, et al. STAT1-induced ASPP2 transcription identifies a link between neuroinflammation, cell polarity, and tumor suppression[J]. Proceedings of the National Academy of Sciences, 2014, 111(27): 9834.
