Extranodal NK/T-Cell Lymphoma, Nasal Type: A Case Series
Jennifer A. Marks, MD1, Zahraa Ghandour, MD2, Victoria Collins, MD2, John Schmieg, MD2, Thomas Atkinson, MD3
1Tulane University School of Medicine, New Orleans, LA
2Department of Pathology, Tulane University School of Medicine, New Orleans, LA
3Department of Hematology and Oncology, Tulane University School of Medicine, New Orleans, LA
Abstract
Extranodal Natural Killer/T-cell lymphoma, Nasal Type is a rare and aggressive Non-Hodgkins Lymphoma associated with the Epstein-Barr Virus and Hemophagocytic Lymphohistiocytosis. We describe three patient presentations at two medical centers in New Orleans, Louisiana, observed within a six-month interval. Although the disease has a lower incidence in the US than in Asia and Latin America, the incidence appears to be on the rise. Due to the spectrum of clinical presentations and low index of suspicion for the disease, presenting signs and symptoms of ENKL may be misattributed to a non-neoplastic condition, resulting in a delay in diagnosis. Increased awareness of the clinical and pathologic spectrum of ENKL, particularly in non-endemic areas, remains an essential means of preventing delay in diagnosis and early initiation of treatment.
Introduction
Mature T-Cell and Natural Killer (NK)-Cell neoplasms are a group of Non-Hodgkins Lymphomas (NHL) comprising a heterogenous collection of lymphoid malignancies.1 Both nodal and extranodal lymphomas are classified within this category of peripheral T-cell lymphomas and include Epstein-Barr Virus (EBV)-associated T-cell and NK-cell lymphomas. The most common of the EBV-related NK and T-cell lymphomas is Extranodal NK/T-cell Lymphoma, Nasal Type (ENKL), an aggressive NHL often involving the upper aerodigestive tract.1,2 This malignancy presents almost elusively extranodally.2
ENKL affects the nasal cavity, typically as a destructive, midline lesion with nasal discharge or nasal obstruction with or without epistaxis.1,2 Clinical manifestation may be confused with sinusitis or other benign, non-neoplastic entities.1 Seventy-five percent of patients have localized disease at diagnosis; less frequently, others have disseminated, non-nasal site involvement on initial presentation.2-4
The incidence of ENKL comprises less than 1% of all NHL in the United States and Europe. However, this cancer is on the rise per comparative data from 2016.5 Epidemiologically, the incidence is higher in those of Latin American and Asian descent compared to their Western American counterparts, on the order of 7-10%.6-8
Clinical presentations vary by geography as well as ethnicity.1,6-8 Most cases in the US or Europe have a uniform disease, limited to the nasal cavity, compared to Asian and Latin Americans, who are more likely to have non-nasal, heterogeneous disease at presentation.2,7,8 Hispanic and Asian-Pacific Islanders present with mostly locoregional disease, where only a quarter of patients have distant disease at diagnosis; non-Hispanics have localized and metastatic disease at roughly similar frequencies.2,6-8
Aggressive NK and T-cell neoplasms are not uncommonly associated with the syndrome of Hemophagocytic Lymphohistiocytosis (HLH), a condition of immune dysregulation and hyperinflammatory response often triggered by infection such as EBV, rheumatic disease, or other malignancy.9,10 This immune response is mediated by massive cytokine production, engulfment of hematopoietic cells by activated macrophages, and excessive tissue damage. The diagnosis of HLH is established by fulfilling either a molecular diagnosis or five out of eight well-established clinical or laboratory criteria.11,12
HLH is observed in nearly 3% of cases of ENKL in endemic nations.8 According to a Japanese study, the dual diagnosis portends a median survival time of only one to two months.13-15 While ENKL has a dismal prognosis alone with a median 5-year overall survival of <15%, the survival precipitously worsens when complicated by HLH.14,15 Mortality rates in such cases are >95%.15
In this case series, we describe three patient presentations at two medical centers in New Orleans, Louisiana, observed within a six-month interval.
Case 1
A previously healthy 46-year-old man from Mexico presented in January 2017 with swelling, erythema, and ulceration of the right nose, cheek, and lip. He had a 2-month onset of B-symptoms (fever, drenching night sweats, and weight loss >10 %) in addition to his previous symptoms. Laboratory results showed hyperferritinemia, hypofibrinogenemia, elevated soluble IL-2 receptor, and pancytopenia, in addition to hemophagocytic histiocytosis on bone marrow biopsy with diffuse in situ hybridization for EBV-encoded RNA (EBER) positivity. EBV DNA quantification titers were markedly elevated. A large, black pustular lesion of the right naris failed to respond to antimicrobials, prompting a biopsy. Pathology revealed ENKL and staging by CT and endoscopic evaluation showed stage IIE disease (Figure 1).
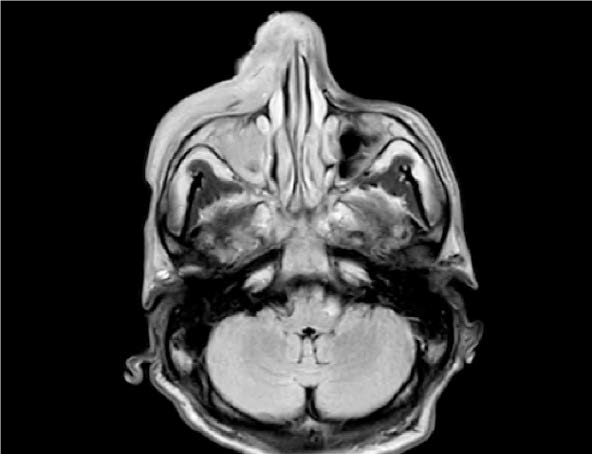
Figure 1: MRI of the Orbit, Face, and Neck, Axial T2 Flair: Diffuse edema of right side of the face and multiple soft tissue enhancing masses on the right side of the nose, along the masseter muscle, soft palate and uvula, and the left tonsillar region, consistent with the clinical impression of lymphoma.
He underwent treatment with concurrent cisplatin with radiation, followed by consolidation chemotherapy with etoposide, ifosfamide, cisplatin, and dexamethasone (VIPD).16,17 He experienced a disease-free interval of approximately eight months. One year later, he relapsed with local recurrence and completed treatment with dexamethasone, methotrexate, ifosfamide, L-asparaginase, and etoposide (SMILE) without complications.18,19
Case 2
A 56-year-old woman with a history of chronic sinusitis presented for outpatient septoplasty in January 2017. She had persistent congestion and crusting of her nasal mucosa with transient epistaxis. Intraoperatively she had a large, irregular, soft tissue mass in the left middle meatus and ethmoid sinus that was biopsied. She was discharged home with an anticipated outpatient follow-up of her biopsy results, but the onset of progressive fatigue, malaise, dyspnea, and altered mentation prompted hospital admission. On evaluation, she was febrile with hepatosplenomegaly and bicytopenia. A bone marrow aspiration and biopsy showed atypical lymphoid infiltration, strongly positive for CD56, perforin, granzyme, CD38, and EBV by EBER. Her marrow was also weakly positivity for CD30 with numerous hemophagocytic histiocytes, concerning for HLH (Figure 2).
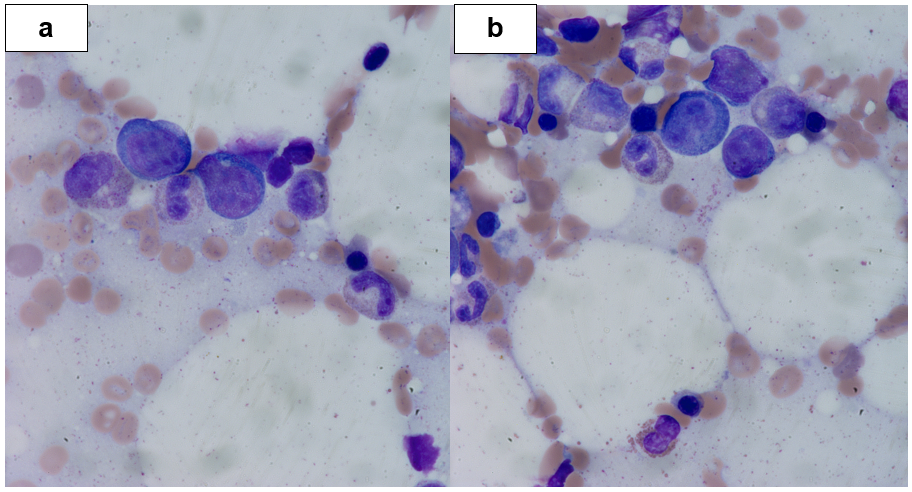
Figure 2: Bone Marrow Biopsy, (a) Left 1000x, (b) Right 1000x: The magnified section does show hemophagocytic histiocytes.
Her CT head and MRI brain were unrevealing, but her surgical pathology was histologically consistent with NK/T-cell Lymphoma.
Given her rapid clinical decline and poor performance status upon hospitalization, she was not a candidate for cytotoxic chemotherapy. She instead received daratumumab, a monoclonal antibody against CD38, in the setting of high expression2,17. Additional workup for HLH was deferred due to the development of multisystem organ failure and clinical decline. She transitioned to comfort care soon after that.
Case 3
A 37-year-old man from the Ivory Coast presented in May 2017 with a one-month duration of fevers, chills, weight loss, cough, and dyspnea. He was acutely hypoxemic on hospital arrival with a CT chest revealing diffuse, bilateral, patchy pulmonary infiltrates (Figure 3). Despite aggressive antimicrobial therapy and supportive measures, his respiratory status worsened, requiring intubation. He underwent bronchoscopy with biopsy. Pathology revealed pulmonary involvement with EBV-positive ENKL, diagnostically with NK/T-cell markers on immunophenotyping and by EBER.
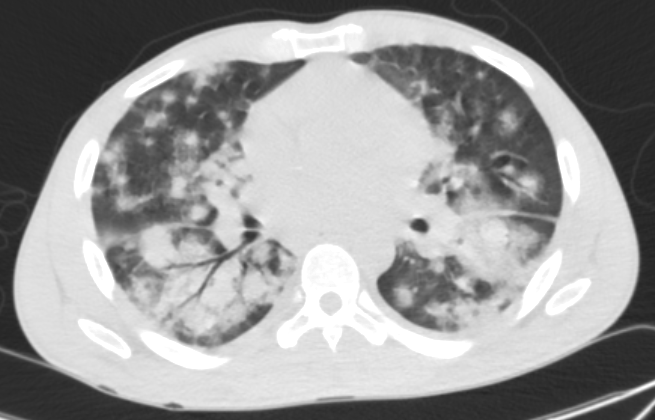
Figure 3: CT Chest: CT of the chest showing diffuse bilateral patchy pulmonary infiltrates with subsequent development of hypoxic respiratory failure requiring intubation.
A bone marrow biopsy was negative for lymphomatous involvement but showed increased histiocytes, including occasional hemophagocytic forms. A CT scan of the sinuses showed right maxillary sinus opacification but no masses. Laboratory studies revealed hyperferritinemia, elevated soluble IL-2 receptor, and pancytopenia, with a constellation of findings consistent with a clinical diagnosis of HLH secondary to ENKL. He remained in critical condition, and his HLH was not expected to improve without treatment of his underlying lymphoma. He started treatment with dexamethasone, methotrexate, ifosfamide, L-asparaginase, and etoposide (SMILE) for Stage IVE disease, which was discontinued early due to the development of transaminitis. Unfortunately, less than a month after his presentation, he clinically deteriorated. He went into pulseless electrical activity arrest without successful return of spontaneous circulation.
Post-mortem analysis was quite dramatic, with evidence of ENKL involvement in numerous organs, including the myocardium pictured below (Figure 4).
Figure 4: Left Ventricle, (a) Left 10x, (b) Right 20x: Sections of the left ventricle demonstrate focal involvement by the NK/T-cell lymphoma. The myocardium is otherwise unremarkable, free of necrosis, fibrosis, and inflammation.
Discussion
Though ENKL remains a rare entity in the Western Hemisphere, the incidence appears to be on the rise per Surveillance, Epidemiology, and End Results datasets.2,5-8 Moreover, the regional distribution of ENTL is quite distinct, comprising 3–10% of NHL in Asia and South America compared to 1% in Western nations.20 Though definitive genetic characteristics explaining these epidemiologic disparities are yet unknown, they are possibly related to variations in HLA type, EBV strain, and other environmental factors20.
As the presentation of ENKL is heterogenous, increased clinical awareness and consideration of this lymphoma may avoid detrimental diagnosis delays and lead to earlier therapy initiation.21,22 All three cases presented with upper aerodigestive involvement, though only two of the three had nasal signs and symptoms. Our first and second cases are characteristic of ENKL compared to our third case's dramatic, atypical presentation.1,6,8 The second case was incidentally diagnosed during surgical septoplasty in the setting of mimicry of chronic sinusitis.
In general, the pathologic features of ENKL include invasion of lymphoid cells around vascular walls, which is characteristic in nearly half of all patients.23,24 The occlusion of vessels leads to ischemic necrosis that may be difficult to differentiate from chronic inflammation, further highlighting the importance of testing with EBER on tissue.2,24,25 However, rare instances of EBV-negative disease have been reported.2,24,25 Moreover, EBV DNA titers may assist in supporting the diagnosis and identifying those at risk of developing HLH.2,15 Prior studies suggest higher viral titers in ENKL are associated with higher metastatic potential, decreased responses to chemotherapy, and inferior survival.2,16,22,26
The combination of ENKL with synchronous HLH is far more aggressive and is often a fatal, complicating syndrome.2,15 Up to 20% of patients with NHL-related HLH died within a month of their diagnosis.15 With a 5-year survival rate of only 15%, EKTL had a poorer prognosis than other B-cell lymphomas.13-15,26 Retrospective studies of EKTL-associated HLH have a median survival of 15 days with a 96.4% mortality rate, underscoring the disease's quick progression and dismal prognosis.15 Other studies suggest that the Eastern Cooperative Oncology Group (ECOG) score, extra-upper aerodigestive tract, and cutaneous involvement are all associated with the development of HLH.15 EBV acts as a co-trigger to stimulate NK and T-cells, promoting high tumor necrosis factor-alpha and interferon-gamma levels.15,20 Thus, it is unsurprising that two of the three cases met five of the eight diagnostic criteria for HLH and demised shortly after the diagnosis.15,23
Treatment recommendations vary depending on the sites involved.2,21-26 Early radiation therapy improves overall and disease-free survival and may benefit patients with earlier-staged disease.22 Patients with advanced and recurrent disease typically are treated with concurrent chemoradiation with platinum-based therapy or combination chemotherapies with L-asparaginase as a backbone.2,16 Efforts for autologous hematopoietic cell transplant have been made but are by and large ineffective.2,20
Novel therapies are increasingly used in practice though the optimal treatment strategy remains elusive.2 Immunotherapeutic agents, such as daratumumab, an anti-CD38, have demonstrated efficacy in EKTL.26-28 This monoclonal antibody works via antibody-dependent cellular phagocytosis and antibody-dependent cell-mediated cytotoxicity27. Our patient received daratumumab given high tumor expression on his tissue but unfortunately died of his cancer shortly after. Moreover, new evidence suggests that treating ENTL-associated HLH with anti-PD1 antibody-based regimens may prolong survival, albeit only for 3.5 months.15,29 Management remains challenging; further studies and therapeutics are needed in this clinical space.2,15,20,27
Conclusion
We present three cases of ENKL spanning a broad spectrum of clinical presentations, disease severity, and treatment responses. High clinical suspicion and prompt workup are essential in securing a diagnosis of ENTL, especially in non-endemic areas. Further studies are needed to elucidate these patients' epidemiologic and environmental genomic and molecular variations, both in the US and abroad, to delineate and develop novel therapeutics.20 The risk for rapid decline with synchronous HLH is high, with a dismal prognosis on the order of days, emphasizing a need for early recognition.15 Increased awareness of the clinical and pathologic spectrum of ENKL remains an important means of preventing delayed diagnosis and timely initiation of treatment.
Funding
The authors have no financial disclosures to declare.
Conflicts of Interest
The authors have no conflicts of interest to report.
References
- Swerdlow SH, Campo E, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood, The Journal of the American Society of Hematology. 2016 May 19; 127(20): 2375-90.
- Allen PB, Lechowicz MJ. Management of NK/T-cell lymphoma, nasal type. Journal of Oncology Practice. 2019 Oct; 15(10): 513-20.
- Au W, Gascoyne R, Klasa R, et al. Incidence and spectrum of non-Hodgkin lymphoma in Chinese migrants to British Columbia. British Journal of Haematology. 2005; 128(6): 792-796.
- Haverkos BM, Coleman C, Gru AA, et al. Emerging insights on the pathogenesis and treatment of extranodal NK/T cell lymphomas (ENKTL). Discov Med. 2017; 23(126): 189–199.
- Dubal PM, Dutta R, Vazquez A, et al. A comparative population-based analysis of sinonasal diffuse large Bcell and extranodal NK/T-cell lymphomas. The Laryngoscope. 2014; 125(5): 1077–1083.
- Ai WZ, Chang ET, Fish K, et al. Racial patterns of extranodal natural killer/T-cell lymphoma, nasal type, in California: a population-based study. British journal of haematology. 2012; 156: 626–632.
- Bassig BA, Au W-Y, Mang O, et al. Subtype-specific incidence rates of lymphoid malignancies in Hong Kong compared to the United States, 2001– 2010. Cancer Epidemiology. 2016; 42: 15–23.
- Haverkos BM, Pan Z, Gru AA, et al. Extranodal NK/T cell lymphoma, nasal type (ENKTL-NT): an update on epidemiology, clinical presentation, and natural history in North American and European cases. Current Hematologic Malignancy Reports. 2016; 11(6): 514–527.
- Campo M, Berliner N. Hemophagocytic Lymphohistiocytosis in Adults. Hematology/Oncology Clinics of North America. 2015 Oct; 29(5): 915-925.
- George MR. Hemophagocytic lymphohistiocytosis: Review of etiologies and management. Journal of Blood Medicine. 2014; 5: 69–86.
- Aricò M, Janka G, Fischer A, et al. Hemophagocytic lymphohistiocytosis. Report of 122 children from the International Registry. 1996; 10(2): 197–203
- Henter JI, Horne A, Aricó M, et al. HLH-2004: Diagnostic and therapeutic guidelines for hemophagocytic lymphohistiocytosis. Pediatric Blood & Cancer. 2007; 48(2): 124-131
- Ishii E, Ohga S, Imashuku S, et al. Nationwide survey of hemophagocytic lymphohistiocytosis in Japan. International Journal of Hematology. 2007 Jan; 86(1): 58–65.
- Li N, Zhang L, Liu J, et al. A clinical study of 21 patients with hemophagocytic syndrome in 295 cases diagnosed with nasal type, extranodal natural killer/T cell lymphoma. Cancer Biology & Therapy. 2017; 18(4): 252–256.
- Liu YZ, Bi LQ, Chang GL, et al. Clinical characteristics of extranodal NK/T-cell lymphoma-associated hemophagocytic lymphohistiocytosis. Cancer Management and Research. 2019; 11: 997.
- Makita S, Tobinai K. Clinical features and current optimal management of natural killer/T-cell lymphoma. Hematology/Oncology Clinics of North America. 2017; 31(2): 239–253.
- Yamaguchi M, Miyazaki K. Current treatment approaches for NK/T-cell lymphoma. Journal of Clinical and Experimental Hematopathology. 2017; 57(3): 98–108.
- Liang JH, Wang L, Peter Gale R, et al. Efficacy of pegaspargase, etoposide, methotrexate and dexamethasone in newly diagnosed adanced-stage extra-nodal natural killer/T-cell lymphoma with the analysis of the prognosis of whole blood EBV-DNA. Blood Cancer Journal. 2017; 7(9).
- Yang L, Liu H, Xu XH, et al. Retrospective study of modified SMILE chemotherapy for advanced-stage, relapsed, or refractory extranodal natural killer (NK)/T cell lymphoma, nasal type. Medical Oncology2013; 30: 720.
- Harabuchi Y, Takahara M, Kishibe K, et al. Extranodal natural killer/T-cell lymphoma, nasal type: basic science and clinical progress. Frontiers in pediatrics. 2019 Apr 16; 7: 141.
- Shawabkeh MAA, Sulaiti MA, Sa’ey HA, et al. Nasal type extranodal natural killer/T (NK/T) cell lymphoma presenting as periorbital cellulitis: a case report. American Journal of Case Reports. 2016 Sep; 17: 934–938.
- Tse E, Kwong Y-L. The diagnosis and management of NK/T-cell lymphomas. Journal of Hematology & Oncology. 2017; 10(1).
- Foucar K, Orazi A. Extranodal Lymphomas and Lymphoid Hyperplasias of Waldeyer Ring, Sinonasal Region, Salivary Gland, Thyroid Gland, Central Nervous System, and Other Less Common Sites. In: Knowles Neoplastic Hematopathology. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2014.
- Kasper DL, Harrison TR. Less common hematologic malignancies. In: Harrison’s Principles of Internal Medicine. 19th ed. New York: McGraw-Hill Education; 2015.
- Jia J, Song Y, Lin N, et al. Clinical features and survival of extranodal natural killer/T cell lymphoma with and without hemophagocytic syndrome. Annals of Hematology. 2016 Jun; 95(12): 2023–2031.
- Wang L, Wang H, Li P-F, et al. CD38 expression predicts poor prognosis and might be a potential therapy target in extranodal NK/T cell lymphoma, nasal type. Annals of Hematology. 2015; 94(8): 1381–1388.
- Wang L, Li LR, Zhang L, et al. The landscape of new drugs in extranodal NK/T-cell lymphoma. Cancer Treatment Reviews. 2020 Sep 1; 89.
- Wang H, Fu BB, Gale RP, et al. NK-/T-cell lymphomas. 2021 Sep; 35(9): 2460-8.
- He Y, Gao Y, Ping L, et al. The emerging role of anti-PD-1 antibody-based regimens in the treatment of extranodal NK/T-cell lymphoma-associated hemophagocytic lymphohistiocytosis. Journal of Cancer Research and Clinical Oncology. 2022 Jul 9: 1-1.
