Chlorine dioxide as a possible adjunct to metabolic treatment
Laurent Schwartz
Assistance Publique des Hôpitaux de Paris, Avenue Victoria 75003 Paris, France
Abstract
A first patient with metastatic adenocarcinoma of the pancreas has decided, on his own, to refuse chemotherapy but to treat himself with lipoïc acid, hydroxycitrate combined with oral ingestion of chlorine dioxide. His blood tests and radiological examinations have almost normalized and the disease is stable at 18 months. Another patient with hormone resistant metastatic prostate cancer has experienced a sharp drop in PSA level as well as improved medical condition. From extensive literature review, the mechanism of action of chlorine dioxide is unknown. It is our hypothesis (albeit unproven) that chlorine dioxide results in tumor cell acidification of the alkaline pH of cancer cells.
Introduction: Cancer is a fermentation process
In the early 1920’s Otto Warburg demonstrated a unique feature of cancer cells, namely an increased uptake of glucose and secretion of lactic acid by cancer cells, even in the presence of oxygen (e.g. the aerobic glycolytic phenotype)1,2. This aerobic fermentation is the signature of cancer3. Warburg also noticed a concomitant decreased number of mitochondria (grana),sup>4
. In normal, differentiated cells, the yield of a molecule of glucose is 34 ATP. ATP is derived mostly from oxidative phosphorylation which takes place in the mitochondria5,6. In the absence of mitochondria the energy yield drops to two molecules of ATP per molecule of glucose5,6. As stated by Warburg in the 1920’s, in cancer cells there is a decreased efficacy of the mitochondria resulting in lesser yield. Despite increased glucose uptake, there is a 50% drop in ATP level in human colon cancer cells compared to adjacent benign cells7. This decrease in ATP is a consequence of impairment of the oxidative phosphorylation6–9.
To compensate for the decreased energy yield, the cell increases its glucose uptake7,10. The decreased activity of the mitochondria has many consequences, one of which is an increased secretion of lactic acid and another one is the activation of the pentose phosphate pathway (PPP). Another consequence is the activation of the glutaminolysis which is necessary for nucleic acid synthesis6–9.
The activation of the Pentose Phosphate Pathway results from an increase in glucose uptake with a concomitant obstacle downstream of the pentose phosphate shunt, most probably at the level of pyruvate dehydrogenase and/or of pyruvate kinase2,6,11. The increased flux in the pentose phosphate pathway results in:
• A shift toward anabolism due to increased synthesis of NADPH that plays a crucial role in NADPH/NADP+ ratio that determines the redox state of the cell via removal of reactive oxygen species (ROS) and so prevents cellular death and controls cellular fate7,11.
• The shift toward the pentose pathway also results in the production of ribose-5-phosphate, required for the synthesis of nucleic acids5.
One other crucial consequences of the mitochondrial defect is intracellular alkalosis7. Tumors show a 'reversed' pH gradient with a constitutively increased intracellular pH that is higher than the extracellular pH. This gradient enables cancer progression by promoting proliferation, the evasion of apoptosis, metabolic adaptation, migration, and invasion12–15.
There is evidence that an acidic extracellular pH promotes invasiveness and metastatic behaviour in several tumor models14,16, proteolytic enzyme activation and matrix destruction17–19.
In normal cells, the intracellular pH (pHi) oscillates during the cell cycle between 6.8 and 7.37. The oscillation of the pH during the cell cycle matches the value of the decompaction of the histones, RNA polymerase activation, DNA polymerase activation and DNA compaction before mitosis7,11.
The intracellular pH of the cancer cells has been less studied. During the cell cycle, it oscillates between 7.2 and 7.5. Intracellular alkalosis is probably a consequence of the decreased oxidative phosphorylation resulting in decreased secretion of carbon dioxide (CO2) and the CO2 reacts with water to create carbonic acid. Cell transformation or enhanced cancer cell division and resistance to chemotherapy are all associated with a more alkaline pHi20–23.
The Warburg effect may be a direct consequence of the activation of oncogenes6. Infection by an oncogenic virus or exposure to a carcinogen inhibits the mitochondrial function and causes the Warburg’s effect24–29.
Reversing the Warburg inhibits tumor growth
The introduction of normal mitochondria into cancer cells restores mitochondrial function, inhibits cancer cell growth and reverses chemoresistance30-35. Also the fusion of cancer cells with normal mitochondria results in increased ATP synthesis, oxygen consumption and respiratory chain activities together with marked decreases in cancer growth, resistance to anti-cancer drugs, invasion, colony formation in soft agar, and « in vivo » tumor growth in nude mice31.
As the Warburg aerobic glycolytic phenotype and its effects on metabolism are key to cancer, the obvious question is whether drugs can be designed to target it. To alleviate the Warburg effect, pyruvate should be converted into Acetyl-CoA, which would decrease the bottleneck that results in the activation of both the Pentose Phosphate Pathway and the glutaminolysis. The mitochondrial yield should be increased to stimulate the synthesis of CO2 and the increased secretion of CO2 would result in a decreased intracellular alkalosis.
The combination of α-lipoïc acid and hydroxycitrate36–39 has been reported to slow cancer growth, in murine xenografts. This inhibition appears to be independent of the primary tumor site and has been reproduced in different laboratories40,41.
The most likely mechanism of action for α-lipoïc acid in its reduction of tumor growth is the inhibition of pyruvate dehydrogenase kinase (the same target of Dichloroacetic acid (DCA)). This enzyme inhibits the activity of pyruvate dehydrogenase and is known to be up-regulated in cancer cells expressing the Warburg aerobic glycolytic phenotype. Pyruvate dehydrogenase catalyses the conversion of pyruvate to acetyl-CoA, the initial step of the final conversion of glucose to carbon dioxide and water in the TCA cycle, with the concomitant production of ATP. Therefore, it is reasonable to suggest that blocking the activity of pyruvate dehydrogenase kinase will at least partially restore the activity of pyruvate dehydrogenase, thereby increasing the flux of pyruvate through the TCA cycle in the mitochondria, while simultaneously reducing the production of lactic acid and most importantly decreasing the flux in the pentose pathway shunt9.
There are several reports of metabolic treatment utilizing a combination of α-lipoïc acid and hydroxycitrate together with conventional cancer therapy. Starting in January 2013, metabolic treatment (α-lipoïc acid/hydroxycitrate with low doses of chemotherapy plus Naltrexone) was offered to patients sent home after the failure of conventional cytotoxic chemotherapy for metastatic cancer (irrespective of the primary site) but with a Karnovsky performance status above 70 (quantification cancer patients' general well-being and activities of daily life)42–46. Of the first randomly selected eleven patients, five were alive and reasonably well 30 months after the start of treatment43-45.
In the update of a subsequent study, patients with multiple brain metastasis (n=4) or glioblastoma (n=6) were treated with a combination of conventional and metabolic treatments (α-lipoïc acid/hydroxycitrate) as well as ketogenic diet. Five out of six patients with glioblastoma were alive and stable after two years, while two of the four patients with multiple brain metastases are alive and well three years later46,47.
Cases reports:
Patient number 1
The first case is a 65 year old French gentleman with biopsy-proven unresectable well differentiated adenocarcinoma of the pancreas in June 2016 . The cholestasis was treated by a derivation in December 2016 which was effective in alleviating the obstruction. The tumor markers (CA 19-9 and CEA) were uninformative. The patient subsequently refused chemotherapy and decided by himself to start a treatment involving
1) Ketogenic diet
2) Lipoïc acid 800 mg twice a day and hydroxycitrate 500mg three times a day
3) Chlorine dioxide up to 32 drops per day. Chlorine dioxide was produced by the activation of NaCLO2 by 4% HCl. The activation time takes 3mn and a drop contains around 86 micromoles of CLO2 if the activation is total.
As of 9/2017, the patient was living normally, the blood tests were normal, the tumour mass such as seen on CT scan had grown from 3 to 5 cm. No side effects were noted. There was ne concomitant chemotherapy or radiation therapy.
Patient number 2
Having heard of the first case, a second patient started a similar treatment. He is a 67-year-old man. He was diagnosed in 8/16 with Gleason 8 adenocarcinoma of the prostate responsible of a cord compression that was successfully treated by laminectomy and post op radiationtherapy. At the start of disease PSA was 1320. Degarelix was started in 8/16 with concomitant ketogenic diet. Because of partial responds chemotherapy with Docetaxel (150mg IV) was started in December 2016. Chemotherapy was discontinued after two cycles because of poor tolerance. Simultaneously, starting In mid November metabolic treatment with lipoïc acid as well as hydroxycitrate. He performs weekly assessment of his PSA.
End of march, the PSA had dropped to 27 and stayed at this value up to beginning of June, but started increasing, in three weeks from 27 to 52 (figure 1). Metastatic pain increased and was responsible of Karnovsky of 70. At that stage the patient started to take chloride dioxide. The PSA dropped linearly for eight weeks to 26. The patient took eight times a day, 344 micromoles of chlorine dioxide. After these eight weeks of decrease, the PSA started to increase in three weeks from 26 to 39. At that stage, metastatic pain, which has almost completely disappeared, was responsible of insomnia. He started to take chlorine dioxide drops not only during the daytime but also every 90 minutes at night.
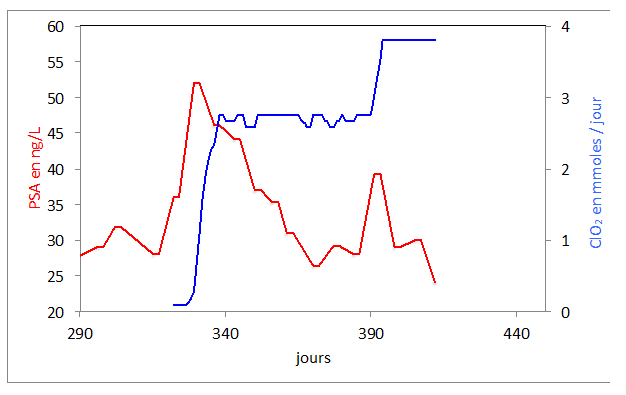
Figure 1: Evolution of the PSA following ClO2 intake in micromole/day in combination with Metabolic treatment.
Nightly metastatic pain decreased drastically from day one, and the second part of the night was practically pain free. The PSA decreased again linearly from 39 to 24.
Discussion and conclusion:
Chlorine dioxide is a poorly studied chemical entity. The mechanism of action of ClO2 is poorly understood. It is our hypothesis that chlorine dioxide decreased the intracellular pH. We are currently running experiments to confirm/infirm this hypothesis. Correcting the intracellular pH may be an alternative or an adjunct to a metabolic treatment as there is extensive literature that many effective cancer treatments decrease the intracellular pH (pHi)2,23,47: literature on increased survival support for the combined use of antacids (which prevent proton extrusion from the tumour cells) with standard chemotherapy15,48–50.
Today, cancer is thought to be a set of very complex diseases with thousands of different mutations. That apparent complexity has led to personalized medicine. However, modern biology has confirmed the universality of the Warburg aerobic glycolytic phenotype. Furthermore, the fact that the combination of α-lipoïc acid and hydroxycitrate slows down cancer growth in every tumor model studied to date suggests that at least some targets are the same in a large spectrum of tumors.
It is possible that the addition of chlorine dioxide increases the response to metabolic treatment.
Acknowledgement:
We want to thank Professor Francis Taulelle for his help in understanding the chemistry and probable mechanism of action of chlorine.
References
- Alfarouk KO. Tumor metabolism cancer cell transporters and microenvironmental resistance. J Enzyme Inhib Med Chem. 2016; 6366: 1–8. doi:10.3109/14756366.2016.1140753.
- Alfarouk KO, Verduzco C, Rauch AK, et al. Glycolysis, tumor metabolism, cancer growth and dissemination. A new pH-based etiopathogenic perspective and therapeutic approach to an old cancer question. Oncoscience. 2014; 1: 777–802. doi:10.18632/oncoscience.158.
- Ward PS, Thompson CB. Metabolic reprogramming: a cancer hallmark even warburg did not anticipate. Cancer Cell. 2012; 21: 297–308. doi:10.1016/j.ccr.2012.02.014.
- Warburg O. On the origin of cancer cells. Science. (1956; 123: 309–14.
- Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science. 2009; 324: 1029–1033. doi:10.1126/science.1160809.
- Israel M, Schwartz L. Cancer: a dysmethylation syndrome, John Libbey Eurotext, 2005.
- Moreira JdaV, Hamraz M. Abolhassani E, et al. The Redox Status of Cancer Cells Supports Mechanisms behind the Warburg Effect. Metabolites. 2016; 6. doi:10.3390/metabo6040033.
- Seyfried TN, Shelton LM. Cancer as a metabolic disease. Nutr. Metab. 2010; 7: 7. doi:10.1186/1743-7075-7-7.
- Israël M, Schwartz L. The metabolic advantage of tumor cells. Mol Cancer. 2011; 10: 70. doi:10.1186/1476-4598-10-70.
- da Veiga Moreira J, Peres S, Steyaert JM, et al. Cell cycle progression is regulated by intertwined redox oscillators. Theor Biol Med Model. 2015; 12: 10. doi:10.1186/s12976-015-0005-2.
- SMazurek S, Boschek CB, Hugo F, et al. Pyruvate kinase type M2 and its role in tumor growth and spreading., Semin. Cancer Biol. 2005; 15: 300–8. doi:10.1016/j.semcancer.2005.04.009.
- Alfarouk KO, Muddathir AK, Shayoub MEA. Tumor acidity as evolutionary spite, Cancers. 2011; 3: 408–414. doi:10.3390/cancers3010408.
- Reshkin SJ, Bellizzi A, Caldeira S, et al. Na+/H+ exchanger-dependent intracellular alkalinization is an early event in malignant transformation and plays an essential role in the development of subsequent transformation-associated phenotypes. FASEB J Off Publ Fed Am Soc Exp Biol. 2000; 14: 2185–2197. doi:10.1096/fj.00-0029com.
- Cardone RA, Casavola V, Reshkin SJ. The role of disturbed pH dynamics and the Na+/H+ exchanger in metastasis. Nat Rev Cancer. 5 (n.d.) 786–95. doi:10.1038/nrc1713.
- Harguindey S, Orive G, Luis Pedraz J, et al. The role of pH dynamics and the Na+/H+ antiporter in the etiopathogenesis and treatment of cancer. Two faces of the same coin--one single nature. Biochim Biophys Acta. nd.; 1756: 1–24. doi:10.1016/j.bbcan.2005.06.004.
- Rofstad EK, Mathiesen B, Kindem K, et al. Acidic extracellular pH promotes experimental metastasis of human melanoma cells in athymic nude mice. Cancer Res. 2006; 66: 6699–707. doi:10.1158/0008-5472.CAN-06-0983.
- Huber V, De Milito A, Harguindey S, et al. Proton dynamics in cancer., J. Transl. Med. 2010; 8: 57. doi:10.1186/1479-5876-8-57.
- Kato Y, Lambert CA, Colige AC, et al. Acidic extracellular pH induces matrix metalloproteinase-9 expression in mouse metastatic melanoma cells through the phospholipase D-mitogen-activated protein kinase signaling. J Biol Chem. 2005; 280: 10938–44. doi:10.1074/jbc.M411313200.
- Montcourrier P, Mangeat PH, Salazar G, et al. Cathepsin D in Breast Cancer Cells Can Digest Extracellular Matrix in Large Acidic Vesicles, Cancer Res. 1990; 50: 6045–6054.
- Gillies RJ, Martinez-Zaguilan R, Martinez GM, et al. Tumorigenic 3T3 cells maintain an alkaline intracellular pH under physiological conditions. Proc Natl Acad Sci USA. 1990; 87: 7414–8.
- Moolenaar WH. Effects of growth factors on intracellular pH regulation. Annu Rev Physiol. 1986; 48: 363–76. doi:10.1146/annurev.ph.48.030186.002051.
- Pouysségur J, Dayan F, Mazure NM. Hypoxia signalling in cancer and approaches to enforce tumour regression. Nature. 2006; 441: 437–443. doi:10.1038/nature04871.
- Alfarouk KOKO, Stock CMCM, Taylor S, et al. Resistance to cancer chemotherapy: failure in drug response from ADME to P-gp. Cancer Cell Int. 2015; 15: 71. doi:10.1186/s12935-015-0221-1.
- Seyfried TN. Cancer as a Metabolic Disease: On the Origin, Management, and Prevention of Cancer, 1st ed. John Wiley & Sons. 2012. doi:10.1002/9781118310311.
- D.M. D’Agostino DM, Bernardi P, Chieco-Bianchi L, et al. Mitochondria as functional targets of proteins coded by human tumor viruses. Adv Cancer Res. 2005; 94: 87–142. doi:10.1016/S0065-230X(05)94003-7.
- Parkin DM. The global health burden of infection-associated cancers in the year 2002. Int J Cancer J Int Cancer. 2006; 118: 3030–44. doi:10.1002/ijc.21731.
- Clippinger AJ, Bouchard MJ. Hepatitis B virus HBx protein localizes to mitochondria in primary rat hepatocytes and modulates mitochondrial membrane potential. J Virol. 2008; 82: 6798–811. doi:10.1128/JVI.00154-08.
- Koike K. Hepatitis B virus X gene is implicated in liver carcinogenesis. Cancer Lett. 2009; 286: 60–8. doi:10.1016/j.canlet.2009.04.010.
- Warburg O. On the formation of lactic acid with growth. Biochem Z. 1925; 160: 307–311.
- Elliott RL, Jiang XP, Head JF. Mitochondria organelle transplantation: introduction of normal epithelial mitochondria into human cancer cells inhibits proliferation and increases drug sensitivity. Breast Cancer Res Treat. 2012; 136: 347–54. doi:10.1007/s10549-012-2283-2.
- Kaipparettu BA, Ma Y, Park JH, et al. Crosstalk from non-cancerous mitochondria can inhibit tumor properties of metastatic cells by suppressing oncogenic pathways. PloS One. 2013; 8: e61747. doi:10.1371/journal.pone.0061747.
- Guénin S, Schwartz L, Morvan D, et al. PP2A activity is controlled by methylation and regulates oncoprotein expression in melanoma cells: a mechanism which participates in growth inhibition induced by chloroethylnitrosourea treatment. Int J Oncol. 2008; 32: 49–57.
- Liang XJ, Finkel T, Shen DW et al. SIRT1 contributes in part to cisplatin resistance in cancer cells by altering mitochondrial metabolism. Mol Cancer Res MCR. 2008; 6: 1499–506. doi:10.1158/1541-7786.MCR-07-2130.
- Wallace KB. Adriamycin-induced interference with cardiac mitochondrial calcium homeostasis. Cardiovasc Toxicol. 2007; 7 101–7. doi:10.1007/s12012-007-0008-2.
- Cullen KJ, Yang Z, Schumaker L, et al. Mitochondria as a critical target of the chemotheraputic agent cisplatin in head and neck cancer., J. Bioenerg. Biomembr. 2007; 39: 43–50. doi:10.1007/s10863-006-9059-5.
- Schwartz L, Abolhassani M, Guais A, et al. A combination of alpha lipoïc acid and calcium hydroxycitrate is efficient against mouse cancer models: preliminary results. Oncol Rep. (2010; 23: 1407–16.
- Schwartz L, Guais A, Israël M, et al. Tumor regression with a combination of drugs interfering with the tumor metabolism: efficacy of hydroxycitrate, lipoïc acid and capsaicin. Invest New Drugs. 2013; 31: 256–64. doi:10.1007/s10637-012-9849-z.
- Zachar Z, Marecek J, Maturo C, et al. Non-redox-active lipoate derivates disrupt cancer cell mitochondrial metabolism and are potent anticancer agents in vivo. J Mol Med Berl Ger. 2011; 89: 1137–48. doi:10.1007/s00109-011-0785-8.
- Abolhassani M, Guais A, Sanders E, et al. Screening of well-established drugs targeting cancer metabolism: reproducibility of the efficacy of a highly effective drug combination in mice. Invest New Drugs. 2012; 30: 1331–42. doi:10.1007/s10637-011-9692-7.
- Schwartz L, Supuran CT, Alfarouk KO. The Warburg effect and the Hallmarks of Cancer. Anticancer Agents Med Chem. 2016.
- Wenzel U, Nickel A, Daniel H. alpha-Lipoïc acid induces apoptosis in human colon cancer cells by increasing mitochondrial respiration with a concomitant O2-*-generation. Apoptosis Int J Program Cell Death. 2005; 10: 359–68. doi:10.1007/s10495-005-0810-x.
- Baronzio G, Schwartz L, Crespi E, et al. Early clinical and toxicological results of a combination of natural glycolysis inhibitors (METABLOCTM) on cancer patients. Biomed Res. 2012; 23: 219–222.
- Schwartz L, Buhler L, Icard P, et al. Metabolic treatment of cancer: intermediate results of a prospective case series. Anticancer Res. 2014; 34: 973–80.
- Schwartz L, Buhler L, Icard P, et al. Metabolic cancer treatment: Intermediate results of a clinical study. Cancer Ther. 2014; 10: 13–19.
- Schwartz L, Gabillet J, Buhler L, et al The addition of chloroquine and metformine to Metabloc induces a rapid drop of tumor markers in advanced carcinoma. Cancer Ther. 2014; 10: 20–27.
- LSchwartz L. Combination of Metabolic Treatment of Aggressive Primary Brain Tumour and Multiple Metastases of the Brain. Cancer Res Oncol. 2016; 2.
- Schwartz L, Seyfried T, Alfarouk KO, et al. Out of Warburg Effect: an effective cancer treatment targeting the tumor specific metabolism and dysregulated pH. In Seminars in cancer biology. 2017; 43: 134-138.
- Cosentini E, Haberl I, Pertschy P, et al. The differentiation inducers phenylacetate and phenylbutyrate modulate camptothecin sensitivity in colon carcinoma cells in vitro by intracellular acidification. Int J Oncol. 2001; 19: 1069–74.
- Hofer KG, Mivechi NF. Tumor cell sensitivity to hyperthermia as a function of extracellular and intracellular pH. J Natl Cancer Inst. 1980; 65: 621–5.
- Harguindey S, Henderson ES, Naeher C. Effects of systemic acidification of mice with Sarcoma 180. Cancer Res. 1979; 39: 4364–4371.
